Abstract
Background
To examine if the metabolic distress after traumatic brain injury (TBI) is associated with a unique proteome.
Methods
Patients with severe TBI prospectively underwent cerebral microdialysis for the initial 96 h after injury. Hourly sampling of metabolism was performed and patients were categorized as having normal or abnormal metabolism as evidenced by the lactate/pyruvate ratio (LPR) threshold of 40. The microdialysate was frozen for proteomic batch processing retrospectively. We employed two different routes of proteomic techniques utilizing mass spectrometry (MS) and categorized as diagnostic and biomarker identification approaches. The diagnostic approach was aimed at finding a signature of MS peaks which can differentiate these two groups. We did this by enriching for intact peptides followed by MALDI-MS analysis. For the biomarker identification approach, we applied classical bottom-up (trypsin digestion followed by LC-MS/MS) proteomic methodologies.
Results
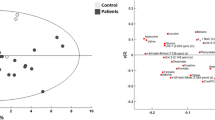
Five patients were studied, 3 of whom had abnormal metabolism and 2 who had normal metabolism. By comparison, the abnormal group had higher LPR (1609 ± 3691 vs. 15.5 ± 6.8, P < 0.001), higher glutamate (157 ± 84 vs. 1.8 ± 1.4 μM, P < 0.001), and lower glucose (0.27 ± 0.35 vs. 1.8 ± 1.1 mmol/l, P < 0.001). The abnormal group demonstrated 13 unique proteins as compared with the normal group in the microdialysate. These proteins consisted of cytoarchitectural proteins, as well as blood breakdown proteins, and a few mitochondrial proteins. A unique as yet to be characterized peptide was found at m/z (mass/charge) 4733.5, which may represent a novel biomarker of metabolic distress.
Conclusion
Metabolic distress after TBI is associated with a differential proteome that indicates cellular destruction during the acute phase of illness. This suggests that metabolic distress has immediate cellular consequences after TBI.








Similar content being viewed by others
References
Marcoux J, McArthur DA, Miller C, Glenn TC, Villablanca P, Martin NA, et al. Persistent metabolic crisis as measured by elevated cerebral microdialysis lactate-pyruvate ratio predicts chronic frontal lobe brain atrophy after traumatic brain injury. Crit Care Med. 2008;36:2871–7.
Bullock R, Zauner A, Woodward JJ, Myseros J, Choi SC, Ward JD, et al. Factors affecting excitatory amino acid release following human head injury. J Neurosurg. 1998;89:507–18.
Vespa M, Prins M, Ronne-Engstrom E, Caron M, Shalmon E, Hovda DA, et al. Increase in extracellular glutamate caused by reduced cerebral perfusion presure and seizures after human traumatic brain injury: a microdialysis study. J Neurosurg. 1998;89:971–82.
Hillered L, Vespa PM, Hovda DA. Translational neurochemical research in acute human brain injury: the current status and potential future for cerebral microdialysis. J Neurotrauma. 2005;22:3–41.
Pineda JA, Wang KK, Hayes RL. Biomarkers of proteolytic damage following traumatic brain injury. Brain Pathol. 2004;14:202–9.
Berger RP. The use of serum biomarkers to predict outcome after traumatic brain injury in adults and children. J Head Trauma Rehabil. 2006;21:315–33.
Haqqani AS, Hutchison JS, Ward R, Stanimirovic DB. Biomarkers and diagnosis; protein biomarkers in serum of pediatric patients with severe traumatic brain injury identified by ICAT-LC-MS/MS. J Neurotrauma. 2007;24:54–74.
Gao WM, Chadha MS, Berger RP, Omenn GS, Allen DL, Pisano M, et al. A gel-based proteomic comparison of human cerebrospinal fluid between inflicted and non-inflicted pediatric traumatic brain injury. J Neurotrauma. 2007;24:43–53.
Gobom J, Nordhoff E, Mirgorodskaya E, Ekman R, Roepstorff P. Sample purification and preparation technique based on nano-scale reversed-phase columns for the sensitive analysis of complex peptide mixtures by matrix-assisted laser desorption/ionization mass spectrometry. J Mass Spectrom. 1999;34(2):105–16.
Larsen MR, Sørensen GL, Fey SJ, Larsen PM, Roepstorff P. Phospho-proteomics: evaluation of the use of enzymatic de-phosphorylation and differential mass spectrometric peptide mass mapping for site specific phosphorylation assignment in proteins separated by gel electrophoresis. Proteomics. 2001;1(2):223–38.
Zuberovic A, Wetterhall M, Hanrieder J, Bergquist J. CE MALDI-TOF/TOF MS for multiplexed quantification of proteins in human ventricular cerebrospinal fluid. Electrophoresis. 2009;30:1836–43.
Hanrieder J, Wetterhall M, Enblad P, Hillered L, Bergquist J. Temporally resolved differential proteomic analysis of human ventricular CSF for monitoring traumatic brain injury biomarker candidates. J Neurosci Methods. 2009;177:469–78.
Lumpkins KM, Bochicchio GV, Keledjian K, Simard JM, McCunn M, Scalea T. Glial fibrillary acidic protein is highly correlated with brain injury. J Trauma. 2008;65:778–82. discussion 782–774.
Zemlan FP, Jauch EC, Mulchahey JJ, Gabbita SP, Rosenberg WS, Speciale SG, et al. C-tau biomarker of neuronal damage in severe brain injured patients: association with elevated intracranial pressure and clinical outcome. Brain Res. 2002;947:131–9.
Afinowi R, Tisdall M, Keir G, Smith M, Kitchen N, Petzold A. Improving the recovery of S100B protein in cerebral microdialysis: implications for multimodal monitoring in neurocritical care. J Neurosci Methods. 2009;181:95–9.
Maurer MH, Berger C, Wolf M, Futterer CD, Feldmann RE Jr, Schwab S, et al. The proteome of human brain microdialysate. Proteome Sci. 2003;1:7.
Brody DL, Magnoni S, Schwetye K, Spinner ML, Esparza TL, Stocchetti N, et al. Science. 2008;321:1221–4.
Marklund N, Blennow K, Zetterberg H, Ronne-Engstrom E, Enblad P, Hillered L. Monitoring of brain interstitial total tau and beta amyloid proteins by microdialysis in patients with traumatic brain injury. J Neurosurgery. 2009;110:1227–37.
Cassimeris L. The oncoprotein 18/stathmin family of microtubule destabilizers. Curr Opin Cell Biol. 2002;14(1):18–24.
Ottens AK, Kobeissy F, Golden E, Ahang A, Haskins W, Chen S, et al. Neuroproteomics in neurotrauma. Mass Spec Rev. 2006;25:380–408.
Yang X, Yang S, Wang J, Zhang X, Wang C, Hong G. Expressive proteomics profile changes of injured human brain cortex due to acute brain trauma. Brain Injury. 2009;23:830–40.
Vespa P, Bergsneider M, Hattori N, Wu HM, Huang SC, Martin NA, et al. Metabolic crisis without brain ischemia is common after traumatic brain injury: a combined microdialysis and positron emission tomography study. J Cereb Blood Flow Metab. 2005;25:763–74.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lakshmanan, R., Loo, J.A., Drake, T. et al. Metabolic Crisis After Traumatic Brain Injury is Associated with a Novel Microdialysis Proteome. Neurocrit Care 12, 324–336 (2010). https://doi.org/10.1007/s12028-010-9342-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-010-9342-5