Article Text
Abstract
Our group has developed a ‘Step Up’ approach to the application of Resuscitative Endovascular Balloon Occlusion of the Aorta (REBOA) in a rural trauma system. This incorporates viewing REBOA as a spectrum of technology. Examples of REBOA technology use to improve outcomes and provision of our system’s clinical practice guideline for the Step-Up application of REBOA technology in the care of trauma patients are presented.
- REBOA
- RAO
- hemorrhagic shock
- trauma
This is an open access article distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited, appropriate credit is given, any changes made indicated, and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0/.
Statistics from Altmetric.com
Introduction
Hemorrhage remains the most prevalent cause of preventable mortality in civilian and military trauma.1–3 Damage control surgery (DCS) has been used by trauma surgeons as a strategy to target life-threatening injuries while delaying all other surgical care until metabolic and physiologic derangements have been corrected. To further improve outcomes in trauma, damage control resuscitation (DCR) has evolved as an extension of DCS.4 5 DCR is a treatment strategy that attempts to maintain oxygen delivery to essential organ systems while mitigating, and if possible avoiding, conditions that exacerbate hemorrhage while source control is achieved. Collectively, contemporary DCR and DCS are most often used together, and advocate early surgical control of hemorrhage and contamination, permissive hypotension until hemorrhage is controlled, and the use of a balanced blood product resuscitation.4 6 7
Importantly, DCR principles and application do not always require surgeons or surgical interventions. The principals of DCR include: (1) hemorrhage control; (2) low volume, permissively hypotensive resuscitation; (3) rapid control of bleeding and contamination; (4) avoiding overuse of crystalloids and colloids; (5) prevention and/or correction of acidosis, hypothermia, and hypocalcemia; and (6) hemostatic resuscitation (early use of a balanced amount of red blood cells, plasma, and platelets or whole blood).4 Research and technology continue to focus on more efficient ways of delivering and assessing the progress of DCR, and one rapidly evolving adjunct is Resuscitative Endovascular Balloon Occlusion of the Aorta (REBOA).8 9
REBOA is the evolution of a concept for hemorrhage control through balloon occlusion of the aorta which was first used in the Korean War.10 Resuscitative aortic occlusion (RAO) via REBOA offers the opportunity to control bleeding in the abdomen and pelvis by endovascular occlusion of the aorta while avoiding the morbidity, potential danger to healthcare providers, and advanced surgical capability associated with or required for direct aortic clamping via resuscitative thoracotomy (RT).11 The current ER-REBOA is able to be inserted using a 7-French introducer sheath (Prytime Medical, Boerne, Texas, USA) that is placed percutaneously. Placement, even in a pulseless patient, is possible if not by ultrasound guidance than by direct vascular exposure and open placement of the sheath.10 A review from two US Level I trauma centers comparing REBOA with RT in critically injured peri-moribund patients demonstrated a higher probability of survival with REBOA; however, in the concept of DCR, a maneuver such as an RT is potentially not an option. Therefore, limiting the REBOA technology to the same indications as RT may not be an equal comparison and could hinder alternative applications.12 Additionally, the concept of placing a REBOA compatible arterial introducer sheath early has been reported to improve survival outcome in critically injured patients regardless of RAO.13
Such reports, and our institution’s experience, support the concept that the spectrum of REBOA technology (eg, early arterial access, arterial 7 French sheath, REBOA catheter, and RAO) improves the care and outcomes of patients in or at severe risk for hemorrhagic shock and cardiovascular collapse that is independent of RAO for hemorrhage control. Presently, guidelines for the use of REBOA and early 7-French introducer sheath placement, as well as outcomes of these maneuvers, remain in evolution. As this newer technology is incorporated into the care of trauma patients, there is the distinct opportunity for misapplication and overutilization, emphasizing the need to address specialty consensus statements regarding who should and should not be placing REBOA.11
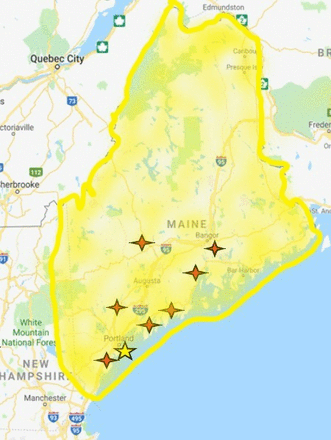
Maine Medical Center (MMC) is the only level I trauma center in the state of Maine and functions as the hub of a rural trauma system that services all of Maine and the majority of Eastern New Hampshire (figure 1). Part of our practice entails the safe application of DCR techniques at our own center and advising referring hospitals on early resuscitation maneuvers prior to transport in this rural trauma system. REBOA was first stocked in the emergency department (ED) at MMC toward the end of 2017 and initially incorporated into routine practice in 2018, and based on our initial use of REBOA technology, we have developed a REBOA oriented treatment paradigm for those in or at significant risk for severe hemorrhage and hemorrhagic shock that we refer to as a ‘Step Up’ REBOA strategy. The step-up concept evolved from literature review and from our institutions initial experience that demonstrated REBOA technology and its potential benefits are not limited to RAO for overt hemorrhage control. We have observed that early arterial access allows continuous blood pressure monitoring and 7 French sheath placement facilitates rapid endovascular treatments for visceral hemorrhage control and intermittent RAO as a method of preferentially diverting blood volume and flow to the heart, lungs, and brain during initial resuscitation (without the need for RT and its associated requirements, morbidity, and risks). Two patients who presented to our institution illustrate this concept.
Rural trauma system map showing the location of Maine Medical Center (yellow star) in regards to additional MaineHealth hospitals (red stars) within the State of Maine. Maine Medical Center’s patient catchment area is highlighted within the shaded yellow region.
Patient 1
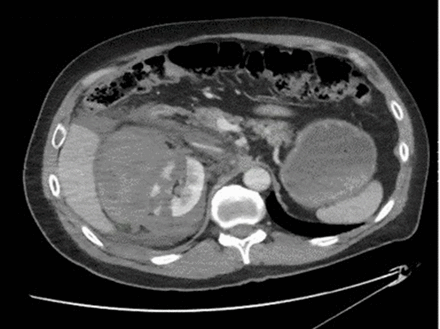
The first patient was a 49-year-old man involved in a helmeted mountain bike accident and self-presented to the ED with a complaint of right flank pain. He was initially stable in the ED and underwent a CT scan of the abdomen and pelvis for flank pain that revealed an American Association for the Surgery of Trauma (AAST) grade 5 renal laceration with active extravasation of contrast coincident with profound hemorrhagic shock (figure 2). Initial resuscitation, allowing for permissive hypotension, included 3 units each of packed red blood cells (pRBC) and fresh frozen plasma (FFP) targeting a systolic blood pressure of 90 mm Hg. The patient had a history of Crohn’s disease with multiple abdominal operations and bowel resections, as well as a ventral hernia repair with mesh. Given the expected difficulty of operative repair, a right common femoral artery (CFA) 7 French arterial sheath was placed under ultrasound guidance and plans made to emergently proceed to surgery for nephrectomy with REBOA use for Zone 1 RAO if needed. However, once arterial access was established with the 7 French sheath, invasive arterial line revealed a higher blood pressure (systolic pressure of 110) than what was noted with noninvasive measurement techniques. The response to resuscitation with the access for immediate REBOA placement and RAO if needed, gave the clinicians confidence and the time to rapidly explore other treatment options in a difficult surgical patient. Urology and Interventional Radiology (IR) were consulted, and in discussions at the bedside, the decision was made to initially pursue angioembolization given the hostile abdomen and successful experience with renal artery embolization, both in renal cell carcinoma and in traumatic hemorrhage.14 15 IR was able to perform the procedure through the Prytime Medical REBOA system’s 7 French sheath which further expedited time to definitive therapy. After embolization, the 7 French sheath was maintained overnight with the REBOA catheter immediately available in the intensive care unit (ICU); neither were needed as the angioembolization proved successful and durable, and the sheath was removed the following morning and the REBOA returned to the REBOA cart. The patient required 3 days in the ICU and was discharged on hospital day 14. Overall, this case illustrated some unappreciated benefits of REBOA technology: (1) Improved monitoring and patient data for triage and medical decision making provided by early arterial access; (2) Emergency medicine and surgeon confidence that RAO could be done in seconds if needed allowing exploration of other management avenues; and (3) IR interventions, including angioembolization, were performed via the already placed 7 French sheath facilitating more rapid therapy in the IR suite. Overall, REBOA catheter insertion for RAO was not ultimately required for the patient to have benefited from the step-up REBOA technology bundle.
CT scan demonstrating a Grade 5 right renal laceration with active extravasation, significant retroperitoneal hematoma and no overt renal pelvis involvement.
Patient 2
The second patient was a 69-year-old woman involved in a high speed car crash where she was rear ended and pushed into oncoming traffic and hit again. She was evaluated at the scene by EMS where she was initially a Glasgow Coma Scale (GCS) score of 13 but had a witnessed ventricular fibrillation arrest, underwent cardiopulmonary resuscitation, two doses of epinephrine, shocked three times, and had bilateral thoracic needle decompression before return of spontaneous circulation (ROSC) was obtained. She was taken to the closest hospital where the needle decompression was replaced with bilateral tube thoracotomies. The initial hospital had access to a limited amount of blood and blood products for transfusion (3 units of pRBC and 2 units of FFP). The patient was given all of this in addition to 1 unit pRBC and 1 unit FFP that Maine Life Flight carried. The patient was sent with emergent aeromedical transport to our hospital for further management in critical condition and extremely tenuous from a hemodynamic standpoint.
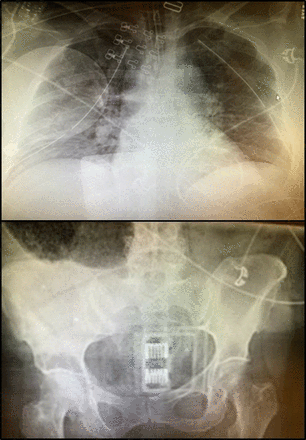
On initial evaluation in our ED, the patient had palpable femoral pulses, with an initial heart rate of 127 and blood pressure of 103/72 and a GCS score of 10T (M6, E3, V1T). Shortly after arrival, she had a second witnessed arrest in our trauma bay with ROSC after a single round of chest compressions and 1 dose of epinephrine. After this, given her overall clinical picture including her prearrest GCS and blunt mechanism, in combination with a negative Extended Focused Assessment with Sonography in Trauma (E-FAST), bilateral chest tubes, and a chest X-ray (figure 3) without significant thoracic hemorrhage, the decision to move to REBOA was made in the midst of the arrest. The Prytime 7 French sheath was placed into the right CFA under ultrasound guidance rapidly and without difficulty and the REBOA catheter inserted. RAO was used intermittently in Zone 3 to support and improve blood pressures (with balloon inflation) and conserve blood flow to the heart, lungs, and brain as the Massive Transfusion Protocol was instituted, and the patient responded hemodynamically. With clear chest wall trauma, lung contusions, negative E-FAST, and a pelvic film with a binder on that did not demonstrate overt pelvic displacement (figure 3) and multiple cardiac arrests of uncertain etiology, our decision was that the patient was better served by, and stable for (with REBOA in and able to provide RAO if needed) CT scan rather than empiric exploratory laparotomy. Imaging revealed numerous rib, spine, and pelvic fractures without signs of vertical shear or open book morphology which would be helped by pelvic binding, external fixation or packing in addition to minimal pelvic hematoma without extravasation of contrast. At that time, the patient had stabilized, although was still requiring vasopressors (vasopressin and norepinephrine), and did not require intermittent RAO any longer. After stabilizing hemodynamically, the patient became responsive and followed commands prior to being moved from the ED to the ICU. Intermittent RAO was also used to minimize further hypoperfusion injury to the myocardium. The patient did have a mildly elevated troponin at 0.69 but a normal echo in the trauma bay once stabilized; overall given the traumatic injury, this is consistent with a blunt myocardial injury. However, given her stabilization and subsequent lack of arrhythmias once resuscitated, the argument could be made that this may also have represented as a type II non-ST-elevation myocardial infarction in the setting of the overall response to injury and acute blood loss anemia.
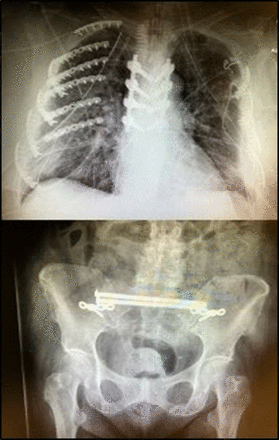
Plain films. Portable chest X-ray demonstrates extensive bilateral rib fractures, bilateral chest tubes and defibrillator pads in place. Portable pelvic X-ray documents empiric pelvic binder in place at arrival.
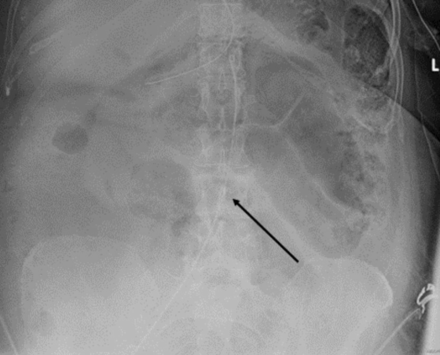
We were ultimately able to appropriately resuscitate the patient in our ICU while leaving the REBOA catheter in place (high Zone 3 but not inflated) overnight (figure 4). Should our resuscitative efforts have failed, we would have been able to immediately gain RAO and proceed to surgery. After stabilization and normalization of her initial metabolic derangements, the patient was able to undergo extensive repair of her injuries (figure 5). The patient required 19 days in the ICU with 12 days on mechanical ventilation and was discharged neurologically intact on hospital day 25. The tone and course of our resuscitation would have been much different if the patient had undergone RT. REBOA allowed us to safely obtain imaging information that steered us toward the ICU and controlled resuscitation and away from surgery which would not have found an obvious source for her hemorrhagic shock. The REBOA system was used not as a bridge to surgery, but as an adjunct to hemostatic resuscitation and facilitated a higher percentage of circulating blood volume to be directed at her heart and brain during the initial resuscitation.
X-ray depicting the placement of the REBOA catheter in Zone 3 (arrow). REBOA, Resuscitative Endovascular Balloon Occlusion of the Aorta.
Films showing postoperative thoracic vertebral and bilateral rib fixation and postoperative pelvic/sacral fixation.
Development of the Step-Up approach to REBOA technology
These two cases illustrate that ‘REBOA’ is not directly dependent on RAO; indeed our initial experience and reviews of the literature led to an appreciation that ‘REBOA’ is a spectrum of technology not specifically dependent on RAO for truncal hemorrhage control to improve outcomes. Indeed, any of the technologies incorporated in REBOA has potential benefits: (1) improved monitoring to guide interventions and direct resuscitation efforts (CFA arterial line for improved hemodynamic monitoring); (2) facilitate non-REBOA interventional therapies (7 French sheath for angioembolization); (3) RAO for conservation/redistribution of limited blood volume to the heart, lungs, and brain while resuscitation occurs, and (4) RAO for truncal hemorrhage control.
These cases demonstrate that a Step-Up approach is effective, and potentially life-saving, and to obtain early femoral arterial access regardless of whether the REBOA catheter is ultimately placed and used. Routine placement of the CFA. A-line also ensures that the training and equipment for arterial lines are in place in the ED. Additionally, if deployed, the REBOA catheter can be beneficial for temporary hemorrhage control and for redistribution of blood volume in the midst of active volume resuscitation. A recent retrospective analysis of REBOA use has indicated potential complications such as increased mortality, acute kidney injury, and limb loss compared with similarly matched controls.16 We are appreciative that some of these can be attributed to access related complications, sheath placement, and by encouraging more access by a broader range of providers that these complications may increase in our population, particularly in the elderly and those with underlying atherosclerotic changes. However, we think that our clinical practice guideline (CPG) can mitigate some of this risk. First, with early placement of smaller gauge arterial line monitors with waveform conformation prior to sheath upsizing, confirmation of CFA placement and associated iatrogenic trauma with immediate sheath placement can be minimized. Second, the smaller 7Fr sheath compared with the previous generation of 12 Fr access for balloon occlusion will lessen the thrombogenic, access, and ischemia-related complications.17 Last, a monitoring protocol with hourly neurovascular checks, bedrest, and minute-to-minute reevaluation of balloon location and necessity of REBOA catheter will keep the duration of intra-aortic catheterization to the minimum, lessen accidental occlusion of renal arteries, and identify patients with thrombotic events early to abate long term sequelae.
As a level I trauma center in a rural system, we care for patients that often arrive well after the Golden Hour’ of initial trauma resuscitation has passed. We considered this in development of our REBOA CPG (see online supplementary file) and appreciate that REBOA is a spectrum of technologies that are potentially best applied in an incremental fashion. We refer to this as the Step-Up approach to REBOA Utilization. This Step-Up approach emphasizes obtaining early common femoral access by placement of an arterial line (18 gauge or larger to facilitate over wire upsizing to a 7 French sheath), which can potentially be done by nonsurgery providers and at transferring centers improving access to the REBOA technology and facilitating stepping up to an arterial sheath or ultimately REBOA catheter if needed. This should provide access to rapid subsequent escalation of REBOA-based interventions (sheath and REBOA catheter placement) and may improve outcomes and proper triage through better monitoring during resuscitation, transport, and more rapid care when the surgeons arrive. We think that this approach can make the spectrum of REBOA technology more available in our own ED and across the spectrum of centers in our catchment area. We are working with PryTime on developing this Step-Up approach and planning its application factoring in the variety of hospital settings and capabilities we work with.
Supplemental material
References
Footnotes
Contributors FRS conceptualized the original idea of the article, developed the concept to CPG, and provided critical revisions and administrative permissions. JPV developed, drafted, and revised the article. JH and JBO contributed to the development of the CPG, article editing, and revisions. NWM contributed critiques and revisions of the CPG along with article editing. CF contributed to article editing, critical revisions, and figures. JR was involved in the institutional compliance of the CPG and article editing. All authors have approved the final version of the article. The corresponding author attests that all listed authors meet authorship criteria and that no others meeting the criteria have been omitted.
Funding The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests JH has the following disclosures: He is a founder and on the Board of Directors of Decisio Health, on the Board of Directors of Zibrio and QinFlow, a Co-inventor of the Junctional Emergency Tourniquet Tool and an advisor to Cellphire Therapeutics, Arsenal Medical and Prytime Medical.
Patient consent for publication Not required.
Ethics approval Maine Medical Center Institutional Review Board Determined as not research.
Provenance and peer review Not commissioned; externally peer reviewed.