Article Text
Abstract
Background Beta-blockers have been proven in multiple studies to be beneficial in patients with traumatic brain injury. Few prospective studies have verified this and no randomized controlled trials. Additionally, most studies do not titrate the dose of beta-blockers to therapeutic effect. We hypothesize that propranolol titrated to effect will confer a survival benefit in patients with traumatic brain injury.
Methods A randomized controlled pilot trial was performed during a 24-month period. Patients with traumatic brain injury were randomized to propranolol or control group for a 14-day study period. Variables collected included demographics, injury severity, physiologic parameters, urinary catecholamines, and outcomes. Patients receiving propranolol were compared with the control group.
Results Over the study period, 525 patients were screened, 26 were randomized, and 25 were analyzed. Overall, the mean age was 51.3 years and the majority were male with blunt mechanism. The mean Injury Severity Score was 21.8 and median head Abbreviated Injury Scale score was 4. Overall mortality was 20.0%. Mean arterial pressure was higher in the treatment arm as compared with control (p=0.021), but no other differences were found between the groups in demographics, severity of injury, severity of illness, physiologic parameters, or mortality (7.7% vs. 33%; p=0.109). No difference was detected over time in any variables with respect to treatment, urinary catecholamines, or physiologic parameters. Glasgow Coma Scale (GCS), Sequential Organ Failure Assessment, and Acute Physiology and Chronic Health Evaluation scores all improved over time. GCS at study end was significantly higher in the treatment arm (11.7 vs. 8.9; p=0.044). Finally, no difference was detected with survival analysis over time between groups.
Conclusions Despite not being powered to show statistical differences between groups, GCS at study end was significantly improved in the treatment arm and mortality was improved although not at a traditional level of significance. The study protocol was safe and feasible to apply to an appropriately powered larger multicenter study.
Level of evidence Level 2—therapeutic.
- brain injury
- propranolol
- randomized controlled trial
This is an open access article distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited, appropriate credit is given, any changes made indicated, and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0/.
Statistics from Altmetric.com
Background
Traumatic brain injury (TBI) is a leading cause of death and disability in our society with over 1.7 million affected annually.1 Limited options exist to prevent the initial or primary brain injury. Secondary injury caused by hypoxia and hypotension may exacerbate the primary injury leading to poor outcomes. Efforts to avoid both factors should be maximized.2 Catecholamine surges that occur in patients with stroke and subarachnoid hemorrhage also occurs in TBI and is often called sympathetic storm, dysautonomia, or paroxysmal sympathetic hyperactivity.3–18 This surge may manifest as hyperdynamic physiologic parameters including tachycardia, hypertension, tachypnea, fever, and paroxysms or any combination of factors.17 18 The magnitude of the surge is often directly proportional to the severity of the TBI.4 19 Few studies have examined the effect of beta-blockade on circulating catecholamines in this patient population.
While the mechanism is unclear, multiple studies in trauma patients have shown the benefit of beta-blockade9–12 14 15 19–23 and several have shown that propranolol (PRO) has improved outcomes compared with other beta-blockers.16 24 25 Despite this compelling evidence, beta-adrenergic blockade is not routine in this population. One recent meta-analysis conditionally recommended beta-blockers in TBI population,23 but routine use is not addressed in most TBI guidelines.26 27
While the preponderance of the evidence is retrospective and observational, enough favorable evidence now exists to justify testing this intervention in a prospective randomized controlled trial. We tested the hypothesis that PRO given to patients with moderate to severe TBI would improve mortality. As a secondary outcome, we examined the effect of treatment with PRO on urinary catecholamines.
Methods
A single-center prospective randomized controlled pilot trial was conducted at the Presley Regional Trauma Center at Regional One Health in Memphis, TN, from January 1, 2016 to December 13, 2017. This hospital is the only trauma center in Memphis, serves as the only trauma center in the Mid-South area, and is a major teaching facility for the University of Tennessee Health Science Center. The Mid-South is a geographic area of approximately 3500 square miles surrounding Memphis including western Tennessee, eastern Arkansas, northern Mississippi, and small portions of Missouri and Kentucky. The trial is registered on ClinicalTrials.gov (NCT02957331). After informed consent was obtained by one of the investigators or research nurses from the patient or legally authorized representative (LAR), patients were randomly assigned to either the treatment (PRO) or control arm using block randomization in groups of 4 to ensure equal group size. This sequence was generated using SAS V.9.4 (SAS Institute). The study was not blinded due to the treatment effects and need for titration of the medication on physiologic parameters.
The primary outcome was in-hospital mortality. The secondary outcome was to examine the interaction between beta-blockade and catecholamines by measuring urinary catecholamines. A power analysis was performed using literature review to determine the probable effect size. Using a 15% difference in mortality between groups, a sample of 99 patients per group would be required to power the study at 0.80. This sample size would not be feasible at a single center. Therefore, in lieu of performing a multicenter study with limited resources, we elected to perform a pilot study to determine the study safety, feasibility, and effect size during a 1-year period. The enrollment was reassessed after 1 year and still short of the goal, so the study was extended an additional year.
Eligible patients for screening were 18 years or older with a TBI as determined by Glasgow Coma Scale (GCS) score less than 12 at admission and documented brain injury on head CT scan. Qualifying patients were randomized within 72 hours of injury. Exclusion criteria included patients with a significant injury in another body region (Abbreviated Injury Scale (AIS) score >3), incarceration or gravidity, hepatic disease, home beta-blocker use, ongoing resuscitation or vasoactive medications, active acute coronary syndrome, and non-survivable injury as determined by neurosurgery review of imaging. Data were extracted from the electronic medical record (Soarian, Cerner, Kansas City, MO) and the local trauma registry (NTRACS V.3.0, Digital Innovations, Forest Hill, MD). Patients assigned to the PRO arm were dosed with PRO starting at 20 mg three times daily by mouth or per feeding tube. The dose of PRO was increased daily in 20 mg three times daily increments (60 mg/day total) until heart rate (HR) was less than 100 beats per minute (bpm) with maximum dose of 640 mg/day. Parameters for holding the medication included HR less than 60 bpm or systolic blood pressure less than 100 mm Hg. Patients assigned to the control arm were managed per institutional standard for TBI based on the Brain Trauma Foundation guidelines.26 Neither arm had additional beta-blockers withheld if the care team deemed that class of medication necessary for appropriate care. Treatment duration for active study intervention was 14 days with a 48-hour taper of PRO after the study period ended in the PRO arm. Patients were followed during the hospital stay until death or discharge.
Variables collected included demographics, severity of injury, severity of illness scores, physiologic parameters, operative interventions, hospital length of stay (LOS), intensive care unit length of stay (ICU LOS), discharge disposition, infectious morbidity, urinary catecholamines, and mortality. Urinary catecholamines were collected from discarded urine at study enrollment and then at study day (SD)-2, SD-5, SD-7, SD-10, and at the end of the study period (SD-14) or when the patient came off study. The samples were stabilized with hydrochloric acid (HCl) and sent to a standard lab. Timely acid stabilization of the urine specimens was required prior to shipping to the central lab for processing. If HCl was not added to the specimen, unpredictable results were obtained that were not physiologically possible in vivo. These values were excluded from analysis. Patients in the PRO arm were compared with the control using intent to treat analysis.
Statistical analyses for categorical variables were compared using χ2 tests or Fisher’s exact tests where appropriate. Normally distributed continuous variables were compared using Student’s t-tests and non-normally distributed continuous variables were compared using Wilcoxon rank-sum tests. Effect size for mortality was calculated using the absolute difference between treatment groups. A two-way mixed model III analysis of variance (ANOVA) with repeated measures was used to estimate differences between the two treatment arms over time. Using planned contrasts, the mean for each follow-up measurement was compared with SD-1 (ie, randomization) for within-group contrasts and means at each assessment time were compared for between-group contrasts. Because these contrasts were planned, no correction was made for multiple comparisons. Output for the ANOVA is reported as least square means with SE (±SE). Finally, Kaplan-Meier survival analysis was performed to further examine the mortality over time between groups. All statistical analyses were performed using SAS V.9.4 (SAS Institute). A p value less than 0.05 was considered significant.
Results
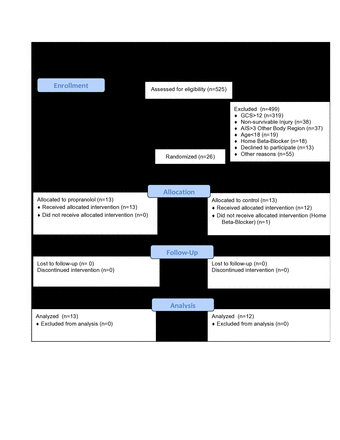
During the 24-month study period, 525 patients were screened for eligibility. Four hundred and ninety-nine patients were excluded from randomization. The majority of the exclusions were GCS score >12 (319) at the time of screening. Additionally, 38 patients had a non-survivable injury, 37 had AIS score >3 in another body region, 19 were less than 18 years old, 18 were on home beta-blocker, 15 were on vasopressors or had ongoing resuscitation, 13 patients declined to participate, 12 were not screened during enrollment window, 9 had no family or LAR available for consent, 8 were incarcerated, 6 had a significant language barrier, 3 were gravid, and 2 had acute coronary syndrome and were excluded prior to randomization. Twenty-six patients were randomized with 13 in each study group. One patient in the PRO group was withdrawn from the study on SD-2 due to home beta-blocker use of which the consenting LAR was unaware at the time of informed consent. The remainder of the patients received assigned treatment according to the study protocol. There was no loss to follow-up and all completed the study protocol during the hospital course or until discharge (figure 1).
CONSORT flow diagram for study population. AIS, Abbreviated Injury Scale; GCS, Glasgow Coma Scale.
Demographics, injury parameters, injury severity, and outcomes are shown in table 1.
Descriptive statistics comparing PRO to the control group
The study groups were comparable with few differences. The injury types were subdural hematoma (72%), subarachnoid hemorrhage (80%), intraparenchymal hemorrhage (28%), epidural hematoma (12%), and diffuse axonal injury (4%) with many patients having combined injuries (72%). Eleven patients (44%) underwent surgical evacuation of hemorrhage with craniectomy (32%) the most common operation performed. The use of intracranial pressure monitors was common (72%) with an intraparenchymal fiber optic monitor (60%) the most frequently used followed by an external ventricular drain (24%). No differences were found with injury types, operative intervention, or type of pressure monitor between study arms.
The median dose of PRO required to achieve HR less than 100 was 60 mg/day until day 12 and then increased to 120 mg/day. The max dose administered was 420 mg/day. Overall, 6% of the doses of PRO were held due to bradycardia or hypotension. Individual patients varied for held doses from none (seven patients) up to 40% of doses held (one patient). Only one adverse event was noted during the study period in a PRO group patient. During the withdrawal period of the PRO, rebound tachycardia occurred which resolved by restarting the medication and using a slower taper. No renal or additional cardiac events, hypotension, or other adverse events were related to study drug. Three patients in the control arm received beta-blockers to control hypertension. All three received the doses of beta-blockers for control of hypertension and none of the three had continuous or scheduled doses. No patients in the treatment arm received any additional beta-blockers.
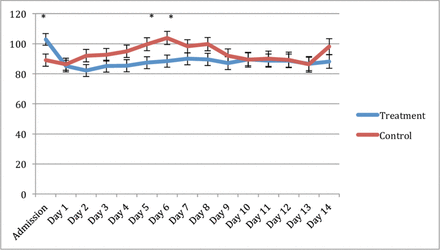
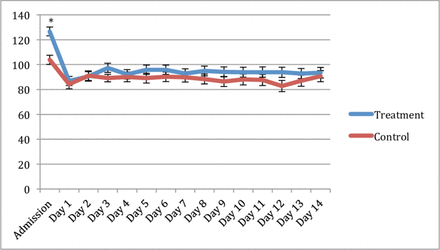
The overall effect of treatment on mean arterial pressure (MAP) was significant with the PRO arm higher (p=0.021). Values with significant differences are highlighted in figure 2. HR was measured from admission to SD-14 and the overall effect of the treatment was not different between arms (p=0.143) (figure 3). No overall treatment differences were found for temperature (p=0.339). No overall treatment differences were found for intra-cranial pressure (ICP).
Least square means (±SE) plot of heart rate (beats per minute) comparing the treatment group (propranolol) to control. *Indicates difference between groups with p<0.05.
Least square means (±SE) plot. Mean arterial pressure (mm Hg) comparing the treatment group (propranolol) to control. *Indicates difference between groups with p<0.05.
The most common overall cause of infectious morbidity was ventilator-associated pneumonia (VAP), which occurred in 68% of the study population. No significant differences were noted for incident VAP, meningitis, bacteremia, or urinary tract infection between groups. No other morbidity occurred related to the study.
The Sequential Organ Failure Assessment and the Acute Physiology and Chronic Health Evaluation II score both improved over the study period. No overall treatment differences were seen between treatment cohorts.
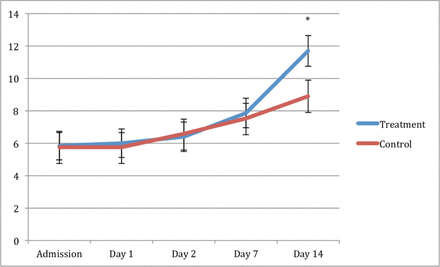
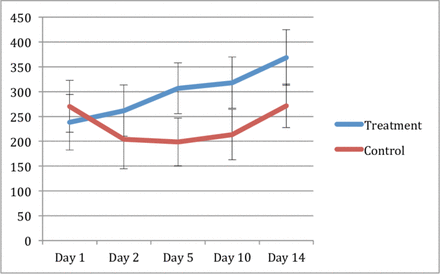
Progression of TBI was examined over the study period with repeated measures of GCS. Both the control and PRO arm had significantly higher GCS at SD-14 as compared with SD-1. The PRO arm GCS was significantly higher at SD-14 as compared with control (11.7 vs. 8.9; p=0.044) (figure 4).
Least square means (±SE) plot of Glasgow Coma Scale score comparing the treatment group (propranolol) to control. *Indicates difference between groups with p<0.05.
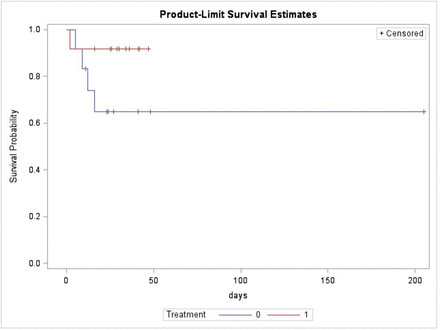
When examining outcomes, no difference was found in mortality between groups. The cause of death was related to the TBI and medical futility in all of the mortalities. There were also no differences detected in LOS or ICU LOS. No differences were detected between groups in overall mortality (7.7% vs. 33%; p=0.109) and survival analysis using the Kaplan-Meier method showed no mortality differences over time (figure 5).
Kaplan-Meier survival analysis. Treatment=propranolol group. No difference between groups with p=0.708.
Overall, 2.4% of the urine specimens were excluded due to lack of stabilizing HCl. After analyzing the remaining specimens, no differences were found in total levels of urine catecholamines over the study period as compared with SD-1 or between study groups (figure 6). Minor differences in the levels of urine dopamine were found at SD-5 and SD-10 compared with SD-1 in the control arm (online supplementary figure 1). No overall treatment differences were found between the groups. No overall differences were found for either urine levels of epinephrine (online supplementary figure 2) or norepinephrine (online supplementary figure 3). Despite not reaching significance at the traditional level, all urinary catecholamines were higher in the PRO arm from SD-2 to end study.
Supplemental material
Supplemental material
Supplemental material
Least square means (±SE) of total urinary catecholamines (μg/L) comparing the treatment group (propranolol) to control.
Discussion
TBI is the leading cause of death and disability in young adults.28–31 Multiple studies have now shown the benefit of beta-blockade in trauma patients9–12 14 15 19–23 25 and PRO may be the most effective medication.16 24 25 A prospective randomized pilot study was undertaken to determine the safety, feasibility, and true effect size of PRO in the TBI population. This study did demonstrate the safety and feasibility of the protocol with only one adverse event during the study period. While we did not demonstrate many significant differences, the study was not powered to detect differences between the study groups. While there was a potentially clinically relevant although not statistically different mortality between groups, all deaths were the result of futile care making the difference difficult to interpret. One other significant finding was that at the end of the study period (SD-14), the GCS in the PRO cohort was significantly higher than the control despite similar initial severity of injury. Even with limited power to the study due to the small sample size, this may indicate a possible beneficial effect to beta-blockade.
Multiple retrospective studies have demonstrated the safety and potential mortality benefit to beta-blockade in patients with TBI. Cotton et al10 examined the effect of beta-blockade on severe TBI with head AIS score >3. Patients receiving two or more doses of beta-blockers had a significant reduction in mortality from 10.8% to 5.1% despite higher injury severity. Our group verified these results and showed that the odds of mortality were reduced with beta-blockers in patients with TBI by 65% in the adjusted regression model.14 These findings are consistent across multiple retrospective studies,9 10 12 14 15 19 20 32 but all share a similar weakness. Patients in most of these studies received ‘2 or more doses’, ‘greater than one dose’, or scheduled doses instead of actually titrating the dose of beta-blockers to a target.10 14 16 24 33 34 This study addressed that weakness by titrating the dose of the PRO to a target HR less than 100. Interestingly, few differences existed in HR between the cohorts and the majority of values are under 100 bmp regardless of group. The true target for HR is unclear, but two studies by Ley et al35 36 suggest outcomes may be improved with more aggressive blockade to HR between 80 and 89.
As the evidence mounts for the protective effect of beta-blockade in TBI, controversy still exists about the best drug to achieve this affect.37 38 Zangbar et al34 performed a propensity matched cohort study with metoprolol compared with no beta-blockers in patients with TBI. One hundred and seventy-eight patients were in each cohort and no differences were found in injury severity, neurosurgical procedures, or HR between the groups. Mortality was decreased by 10% in the metoprolol cohort as compared with no beta-blocker.34 A randomized study using atenolol demonstrated decreased cardiac enzymes, decreased arrhythmias, and T-wave change potentially showing that the end organ effects of the catecholamine surge were blocked.39 Multiple additional studies have shown that PRO increases the protective effect as compared with other beta-blockers. Our group compared PRO with other beta-blockers in patients with TBI and found that the absolute mortality reduction compared with other beta-blockers was 12% and the odds of mortality was reduced by over 80% in adjusted analysis.16 Other authors have confirmed this finding24 33 and a recent prospective observational trial presented at the American Association for the Surgery of Trauma also verified this result showing that the risk of mortality in the PRO cohort was reduced by approximately 50% as compared with other beta-blockers (adjusted OR: 0.51).25 PRO is a lipophilic drug that easily crosses the blood–brain barrier making it an ideal choice in this population.40 Several studies in the murine model have shown a neurologic benefit in surviving animals in a TBI model38 and elucidated a possible mechanism for this benefit. Ley et al37 also used a murine TBI model and showed that the brain perfusion of PRO-treated mice increased by 152% as compared with control with decreased hypoxic areas on immunohistochemistry staining. It is therefore plausible that the PRO may be decreasing central vasoreactivity potentially caused by the catecholamine surge related to the TBI. This suggested effect may lead to better perfusion of the penumbra around the injury and limit potential secondary injury due to decreased oxygen delivery and hypoxia.
The etiology for the syndrome of dysautonomia or paroxysmal sympathetic hyperactivity18 is a catecholamine surge leading to tachycardia, fever, hypertension, diaphoresis, tachypnea, and mydriasis from cerebral disconnection.15 Multiple authors have shown the both central and peripheral catecholamines are elevated after TBI.4 6 13 17 32 41–43 The elevation of catecholamines is directly proportional to the severity of brain injury.4 13 Catecholamine elevation seems to be highest in plasma immediately after injury, but this elevation may persist for up to 14 days.13 32 41–43 In this study, we measured urinary catecholamines and pooled the results for total analysis as well as examined the individual results of dopamine, epinephrine, and norepinephrine. While none of the values were significantly different between treatment groups at the measured time points, the PRO arm was higher for all measurements. This finding is also evident as the MAP was higher in the PRO arm after enrollment, although not significantly so at the traditional level (p=0.052). These results suggest that treatment with PRO may block the end organ effects of the catecholamines and lead to positive feedback further increasing the circulating levels of catecholamines. This study was not powered to show a difference, but the trend is intriguing and clearly warrants a larger multicenter study.
These data must be viewed in light of potential limitations of the study. While we did randomize and perform the trial prospectively, the study is underpowered to show differences between groups. Additionally, selection bias may also be present despite the randomized nature of the trial due to the low enrollment despite screening over 500 patients during 2 years. We could also not blind the trial as the treatment group would be obvious to the caregivers given the effects of PRO on HR and the titration of the medication to HR less than 100.
Conclusion
This trial demonstrated that this protocol is safe and feasible in the TBI population. Despite not being powered to show statistical differences between groups, GCS at study end was significantly improved in the treatment arm and mortality was improved although not at a traditional level of significance. A larger multicenter trial is needed to validate these results and increase the power of the findings.
Acknowledgments
The authors would like to acknowledge Stephanie Baggett and Suzanne Moyer for their contributions to the study. Additionally, the study could not have been completed without the guidance and assistance provided by Lynda Waddle Smith.
References
Footnotes
Contributors Study design: TJS, JPS, CPS, LPC, LJM, ML, MM, JAW, EAT, MAC, TCF. Data collection: TJS, JPS, CPS. Data analysis: TJS, EAT. Data interpretation: TJS, JPS, CPS, MAC, TCF. Drafting of article: TJS, JPS, LPC, MAC, TCF. Critical revision: TJS, JPS, CPS, LPC, LJM, ML, MM, JAW, EAT, MAC, TCF. Final approval: TJS, JPS, CPS, LPC, LJM, ML, MM, JAW, EAT, MAC, TCF.
Funding This study was supported by grants from the University of Tennessee Health Science Center Institute for Research, Innovation, Synergy, and Equity (iRISE) and the Semmes-Murphey Foundation.
Competing interests None declared.
Patient consent for publication Not required.
Ethics approval The University of Tennessee Health Science Center and Regional One Health Institutional Review Boards approved the study.
Provenance and peer review Not commissioned; externally peer reviewed.
Data availability statement All data relevant to the study are included in the article.